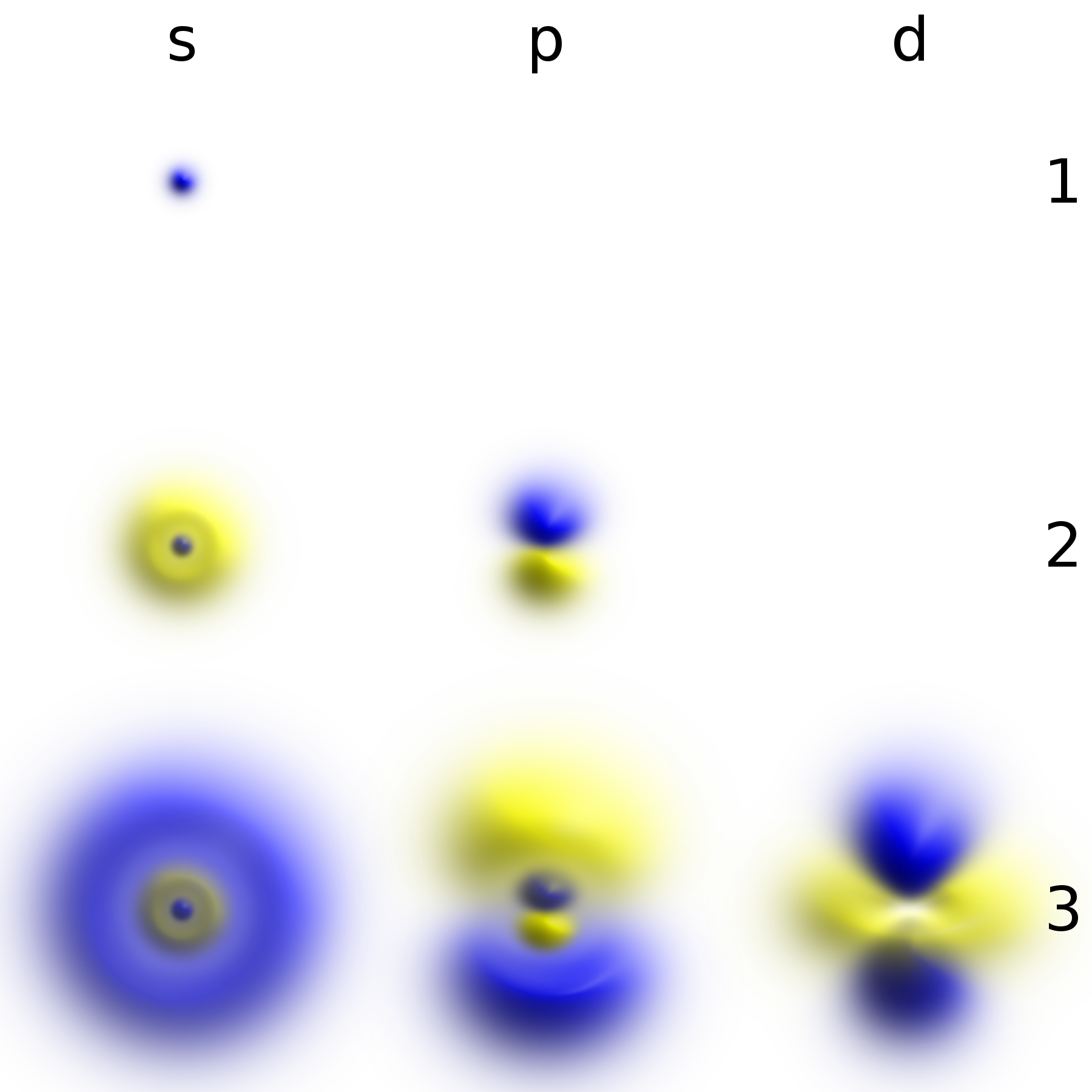
Statistical distribution of the absolute value of electron phase space... | Download Scientific Diagram
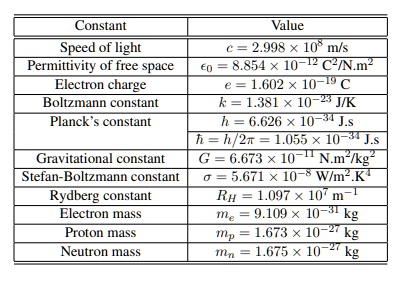
SOLVED: Constant Value Speed of light 2.998 * 10" ms Permittivity of free space 8.854 x 10 C-IN.m Electron charge 1.602 10-19 Boltzmann constant k=1381 * 10– JIK Planck s constant h =

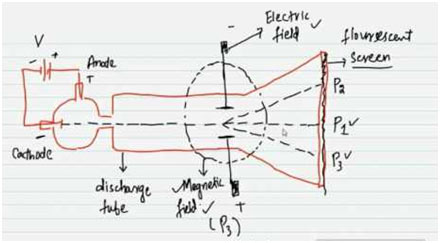
The acceleration of an electron in an electric field of magnitude 50 V/cm, if e/m value of the - YouTube

The charge of an electron is `1.6 xx 10^(-19)C` what will be the value of charge on `Na^(+)` ion. - YouTube

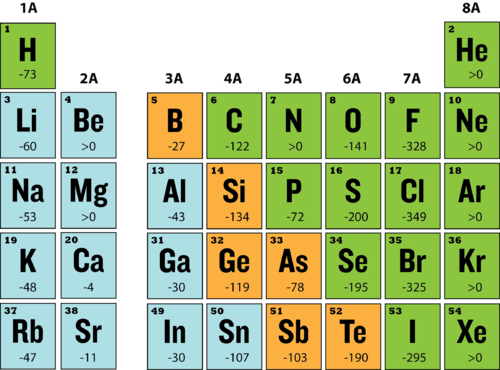
The ' m ' value for an electron in an atom is equal to the number of m values for l = 1 . The electron may be present in