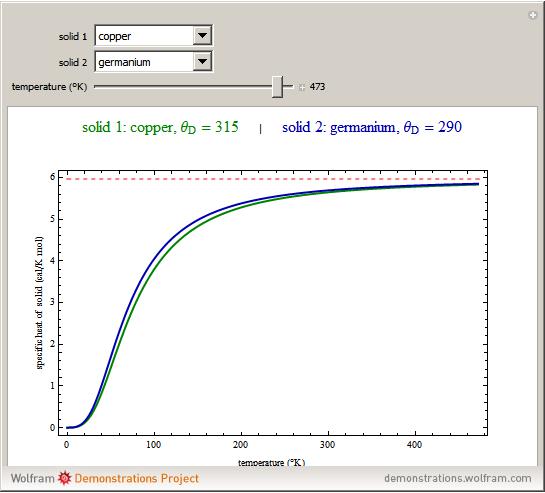
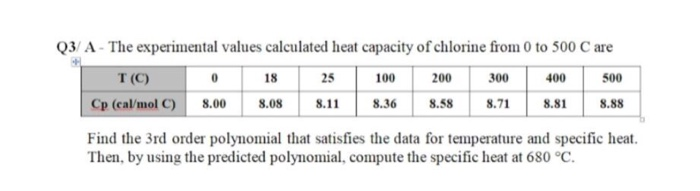
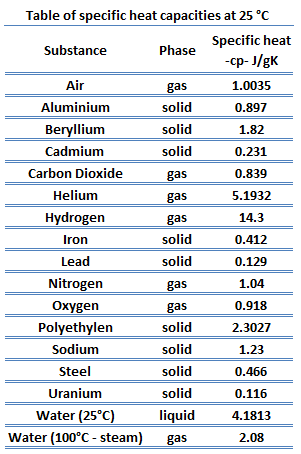
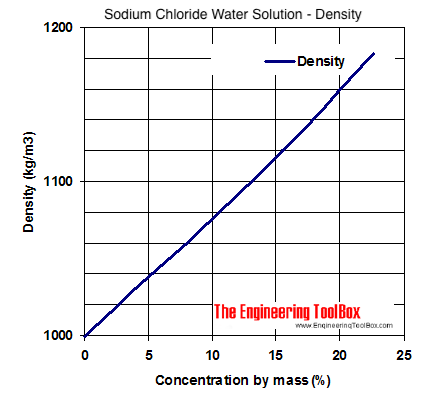
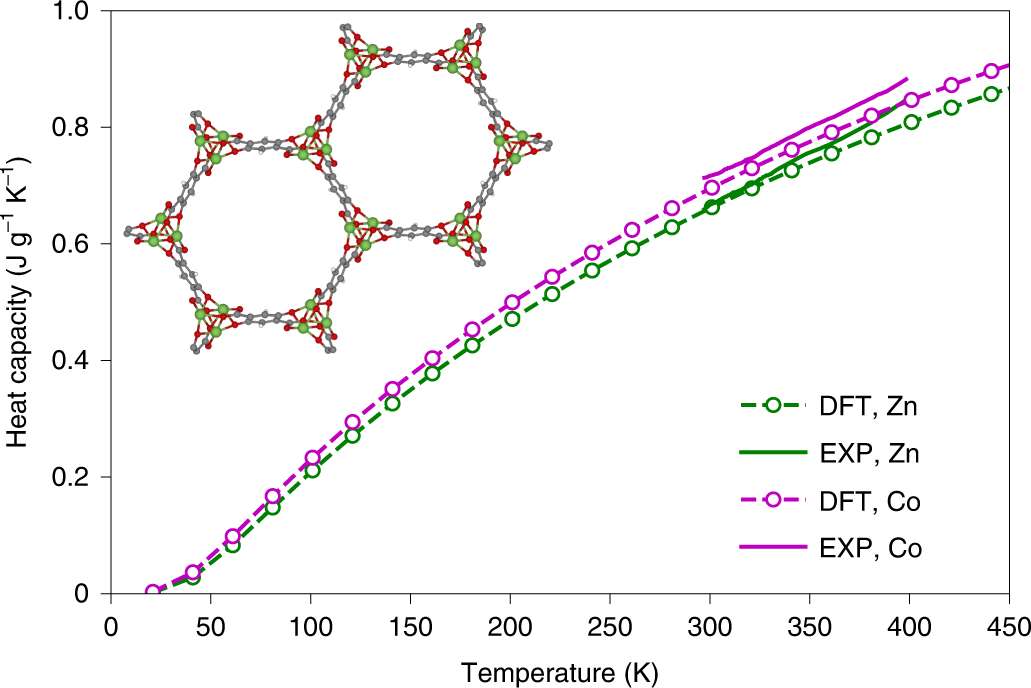
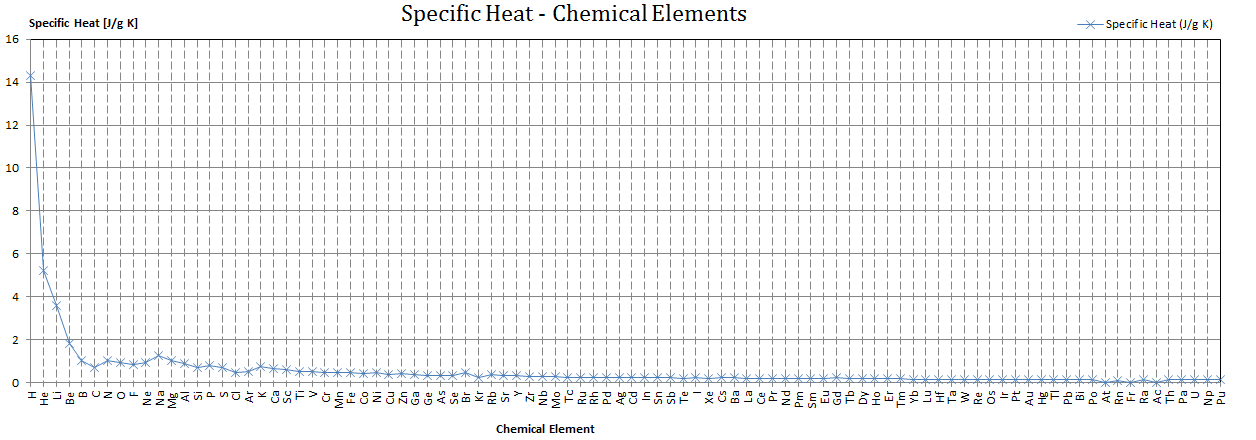
SOLVED: The French chemists Pierre L. Dulong and Alexis T. Petit noted in 1819 that the molar heat capacity of many solids at ordinary temperatures is proportional to the number of atoms
![SOLVED: Q2. Statistical Thermodynamics Answer all parts What do you understand by the term molecular partition function? [10 marks] Write the partition function for a (WO- -level system, the lower state (at SOLVED: Q2. Statistical Thermodynamics Answer all parts What do you understand by the term molecular partition function? [10 marks] Write the partition function for a (WO- -level system, the lower state (at](https://cdn.numerade.com/ask_images/0210408230c14ce4bacfda8c6c3d816b.jpg)
SOLVED: Q2. Statistical Thermodynamics Answer all parts What do you understand by the term molecular partition function? [10 marks] Write the partition function for a (WO- -level system, the lower state (at

Specific heat capacities (C sp ) measured in NaCl, KCl, CaCl 2 , and... | Download Scientific Diagram
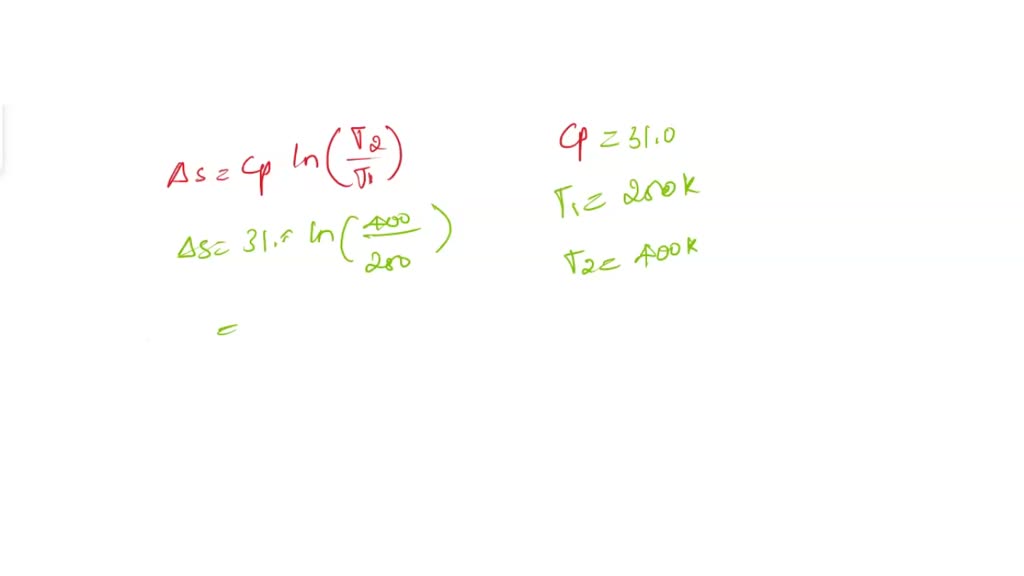
SOLVED: The molar heat capacity of chlorine gas is given by: CP=31.0+0.008 TK(J.K-1.mol-1) Calculate the entropy change when 5 moles of Cl2 are heated from 250. K to 400. K at constant

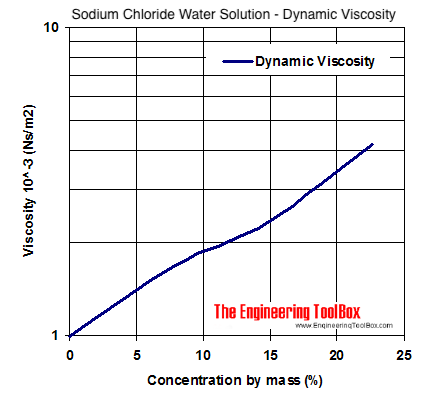
The heat capacity of small particles of sodium chloride - Transactions of the Faraday Society (RSC Publishing)

Specific heat and equivalent wt. of a metal are = 0.057 cal g^-1 and 38.24 respectively. It's exact atomic weight is?
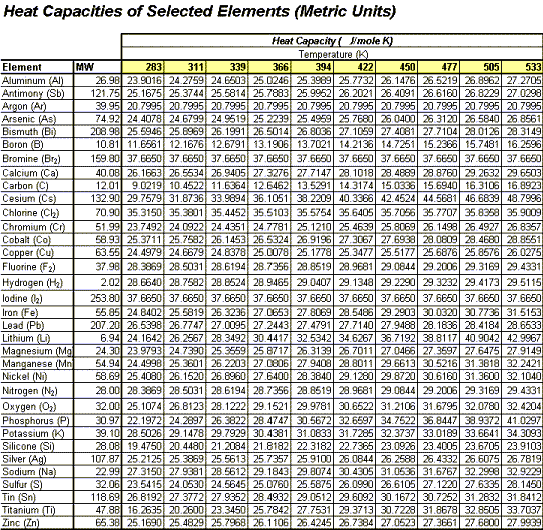
Estimating Heat Capacities for Solutions with Dissolved Solids - Calculations and Tips - Articles - Chemical Engineering - Frontpage - Cheresources.com