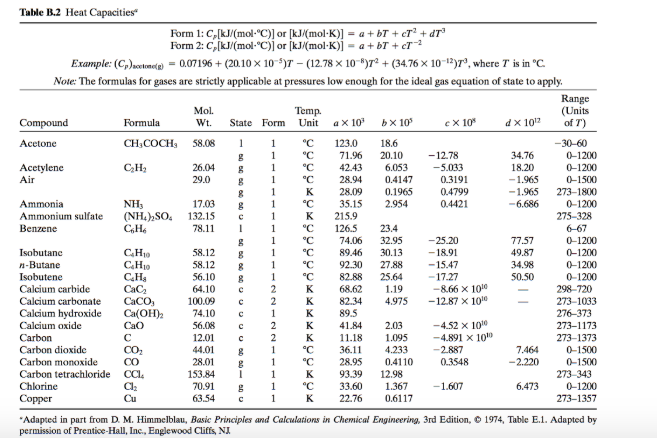
SOLVED: 1. The heat capacity of calcium carbonate (CaCO3, 100.1 g mol–1), is 82.0 J mol–1 K–1. Calculate the energy needed to heat 73.3 g of calcium carbonate from 34.5 °C to

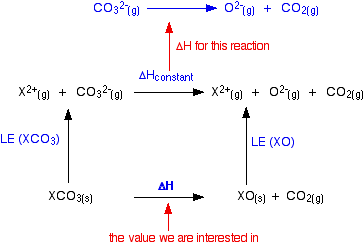
SOLVED: Write the thermochemical equation for the formation of calcium carbonate. The standard molar enthalpy of formation of calcium carbonate is -1207 kJlmol: (2 marks C) How does a catalyst affect the

Toward the Mechanistic Understanding of the Additives' Role on Ammonium Nitrate Decomposition: Calcium Carbonate and Calcium Sulfate as Case Studies | ACS Omega

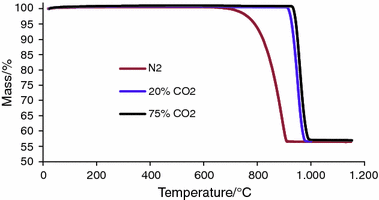
The calcium carbonate (CaCO3) conversion, versus time for different... | Download Scientific Diagram

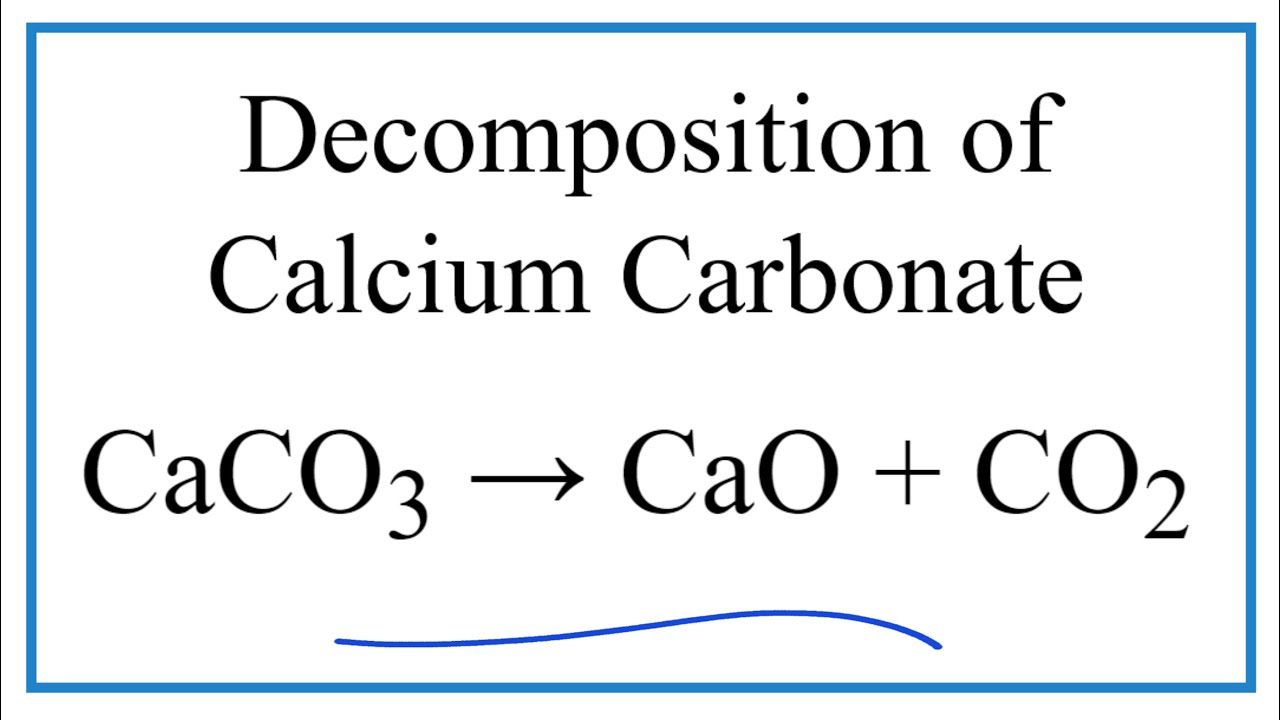
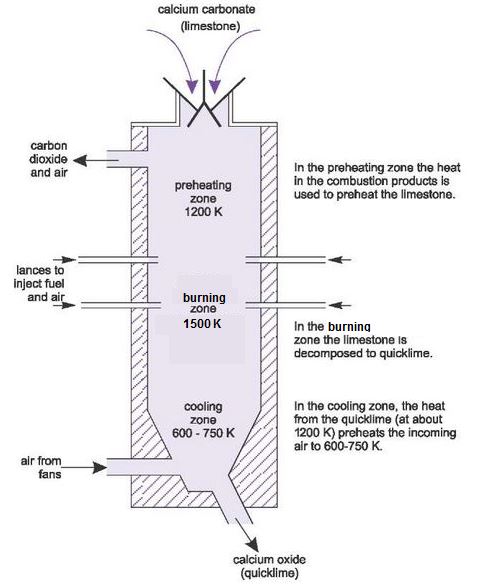
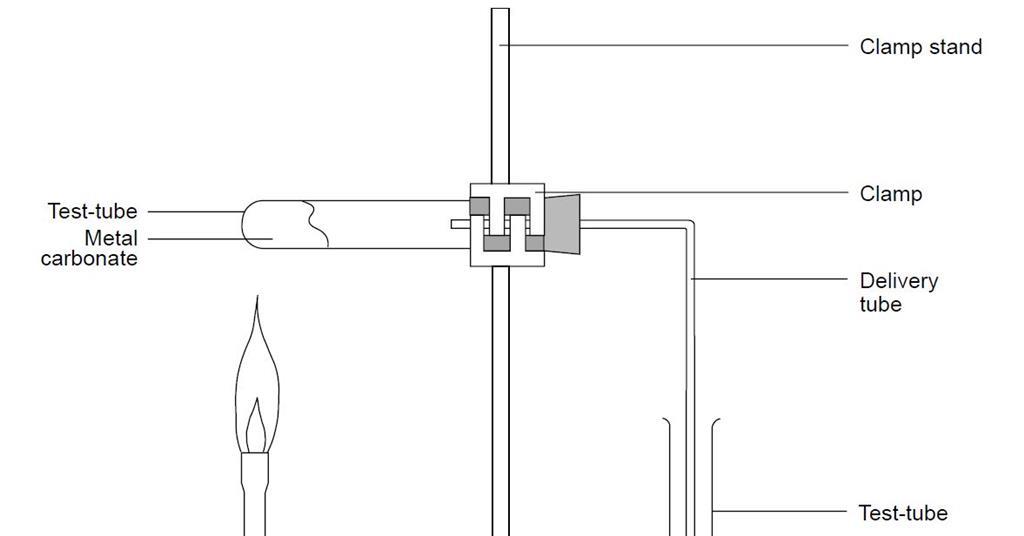
Calcium carbonate decomposes, on heating , to form calcium oxide and carbon dioxide. When 10 g of - YouTube

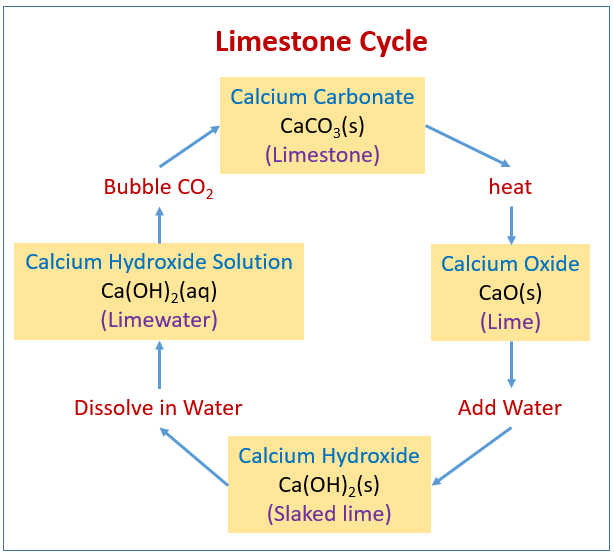
In Denmark the subsoil consists mainly of limestone. In contact with ground water containing carbon dioxide some of the calcium carbonate dissolves as calcium hydrogen carbonate. As a hydrogen carbonate causes problems
![SOLVED: 25. The temperature of a 11.8 g sample of calcium carbonate [CaCO3 (s)] increases from 24.8 -C to 38.0 -€. If the specific heat of calcium carbonate is 0.82 Jlg-K, how SOLVED: 25. The temperature of a 11.8 g sample of calcium carbonate [CaCO3 (s)] increases from 24.8 -C to 38.0 -€. If the specific heat of calcium carbonate is 0.82 Jlg-K, how](https://cdn.numerade.com/ask_images/eedab7cc45b741e085b60c8f5508450c.jpg)
SOLVED: 25. The temperature of a 11.8 g sample of calcium carbonate [CaCO3 (s)] increases from 24.8 -C to 38.0 -€. If the specific heat of calcium carbonate is 0.82 Jlg-K, how




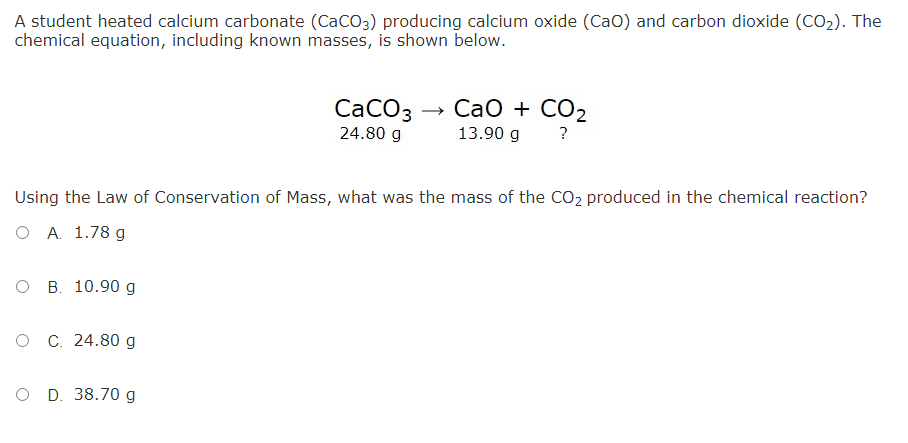





.jpg)