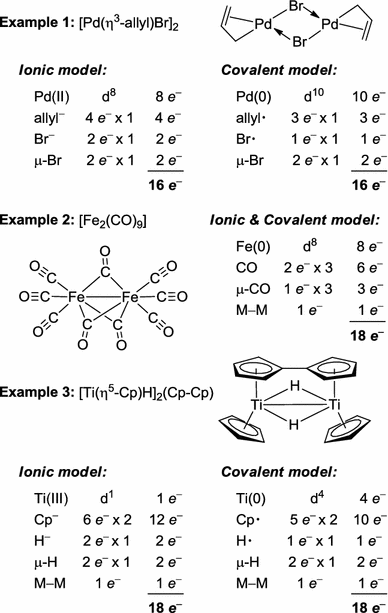
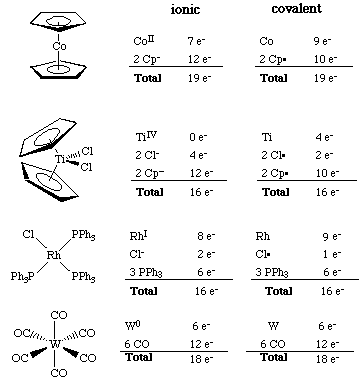
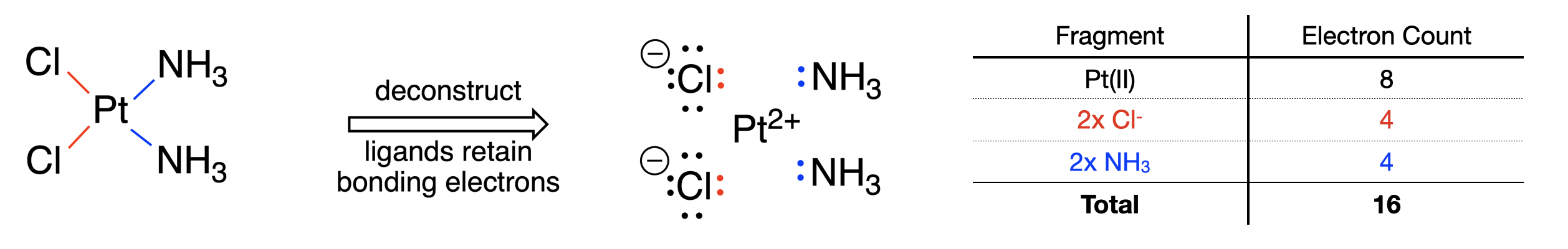
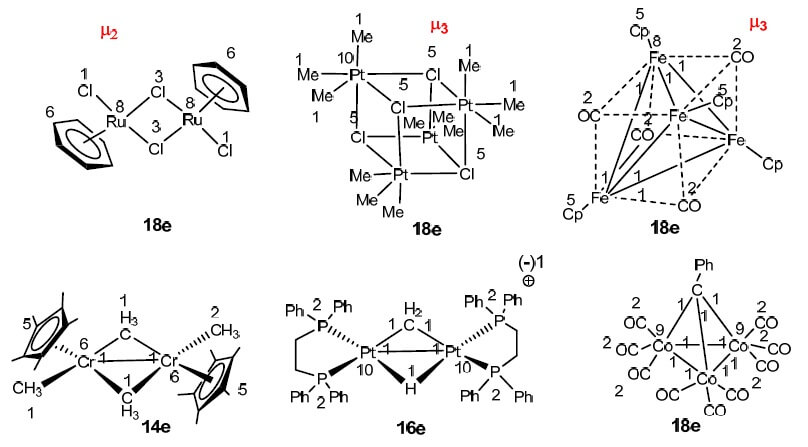
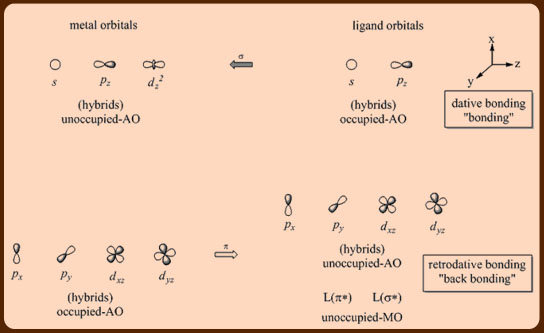
The 16 and 18 electron rule in organometallic chemistry and homogeneous catalysis - Chemical Society Reviews (RSC Publishing)

Using the 18-Electron Rule To Understand the Nominal 19-Electron Half-Heusler NbCoSb with Nb Vacancies | Chemistry of Materials

The realtive stabilities of metal carbonyls and their ions can be explained on the basis of a rule which is known as 18e^(-) rule. If number of valence shell electrons of the
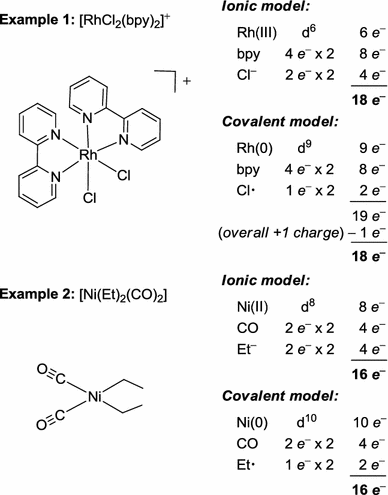
The 18-electron rule and electron counting in transition metal compounds: theory and application | SpringerLink